G
gardenhouse
Experienced
- Mar 26, 2026
- 268
Sorry if this is a repost, This isn't my initial method to CBT, of course if N was available, it would that, but i live in the country with the strictest border force in the western world, my chloroquine bought online from India was seized, I'm sure if I buy SN overseas, they will seize it as well since it's actually available locally but hard to get without proper license.
All kind of SH tools are strictly controlled, I hate this country. So, the only available source locally is NITROGEN, I would like to know if these would work....
I live with my partner, I have no idea where to hide this stuff, hope the gas cylinder is not big....
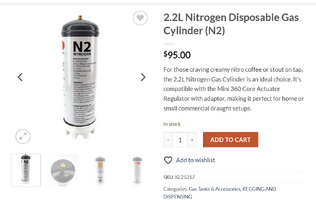

Thanks
All kind of SH tools are strictly controlled, I hate this country. So, the only available source locally is NITROGEN, I would like to know if these would work....
I live with my partner, I have no idea where to hide this stuff, hope the gas cylinder is not big....


Thanks